HAPIGuard - Instructions
HapiGuard – Set up/ Fitting Instructions
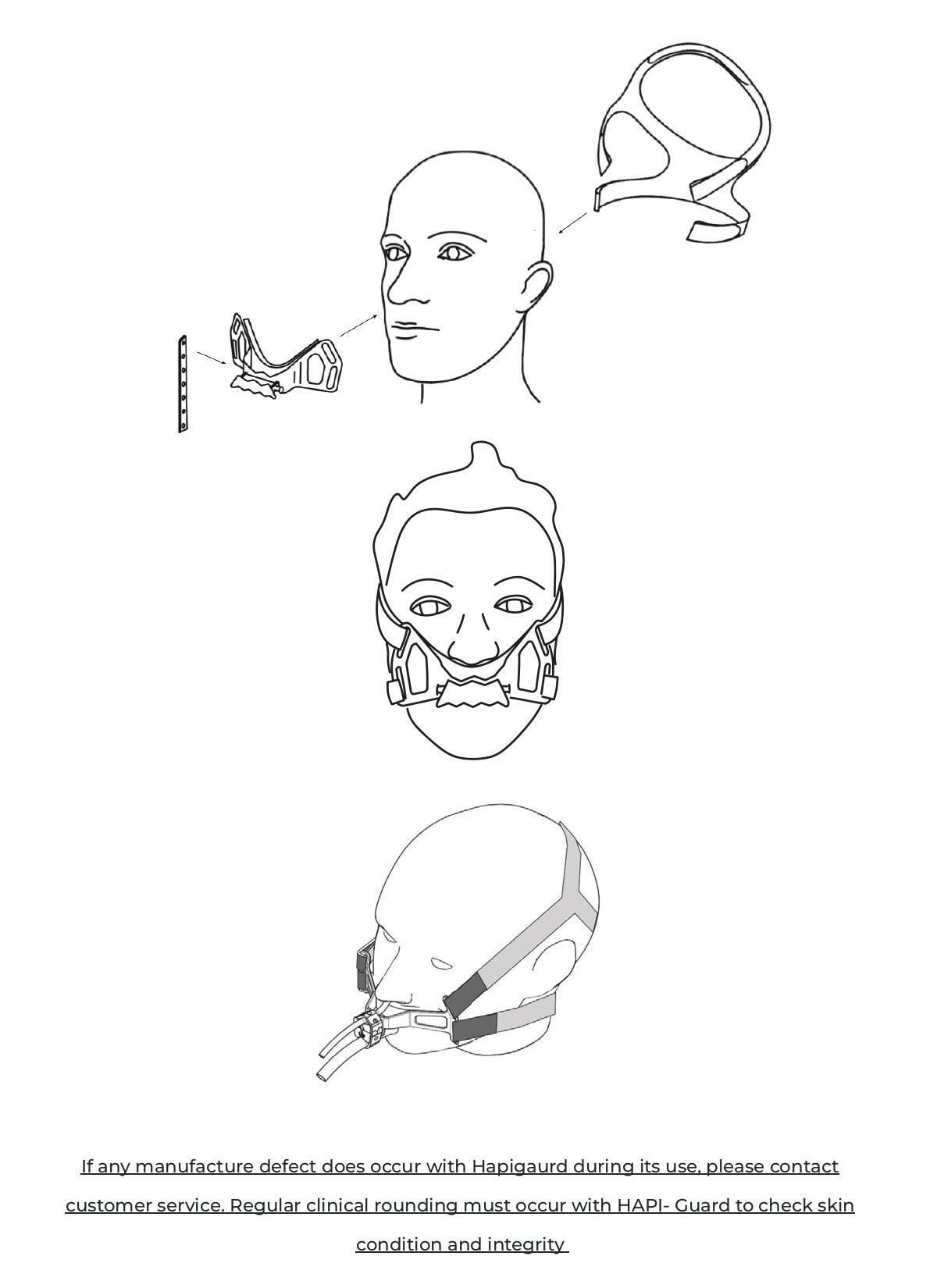
- 1) Remove the three separate components from package (Visual instructions below)
- If there are any questions or concerns with set up or product quality, change HapiGuard or contact customer service.
- Elastic Headband (A “B” will designate the back of head)
- Endotracheal, Orogastric/ Nasogastric Tube Harness (Securement block)
- Silicone strap
- Attach Headband to patient’s head - Straps with Velcro to secure to the back of the neck and at the crown of the head
- 2) Arrow to designate top and bottom position of securement block
- 3) Start from the back of the head, align the straps above and below the ear, loop and secure the Velcro to the top and bottom of the securement block once all four straps are secured to the back of the neck loop the strap at the crown of the head and secure the Velcro, adjust all straps for patient comfort
- 4) Attach Endotracheal, Orogastric/ Nasogastric Tube Harness with the four openings on the harness to the four ends of the strap. The Harness will be placed under, below the patient’s nasal passage/nose. The Harness will be above patients mouth and lips.
- 5) Position ETT to the bottom of the Block grove and secure with the elastic strap position NGT on the top Block grove and secure with the rest of elastic strap
- 6) Attach the silicone strap via silicone eye to the end attachment and wrap around the EET once that continue to connect to the other end attachment. Wrap the strap once on top for NG tubes and connect again to the other end attachment.
- 7) The ETT is expected to be moved from one of the four inserts on the bottom grooves regularly to prevent ETT staying on one part of lips for more that 24 hours.
- 8) If a NG (Above the harness, via Nare, Nasal Collum) tube can be placed in the grove that is associated with the specific Nare the device is placed. This does not need to be moved unless it is no longer needed for the patient.
- 9) If the OG (Below, via Mouth) Tube will attach next to the ETT to secure both at the same time via the silicone strap to the harness.
- 10) If there are Neuro monitoring devices attached to head the Headband will have a separate component that allows to go around the device.
- 11) Please use caution or consult with surgeon and physician with placement concerns prior to attachments
- 12) Please see detail picture for further instructions on placement or contact IMD or IMD salesperso
HAPIGuard - Description & Warnings
1. Description :
- Single Use Patient Care Endotracheal Tube and Orogastric/ Nasogastric Holder. This device comes with three separate components in the package. They are intended to work as one integrated unit which will safely and securely attach ETT and OG/NG tubes to minimize risk of facial device related Hospital Pressure related injuries.
2.Cautions :
- Please inspect product and package prior to use. Do not use product if packaging has been compromised.
3.Indication :
- To prevent Hospital Acquired Pressure device related Injuries and to securely and safely secure ETT and OG/ NG tubes to patients face and head for those patients required airway support and enteral feeding.
4.Contraindications :
-
Hapiguard should never be used for any other purpose than to attach and secure ETT and OG/NG tubes. Hapi Guard strictly prohibits using the ETT or OG/NG for any other purpose than listed above.
Storage of Hapiguard should be between 60-75 degrees during storage and transport.
5.Warnings/ Precautions :
- Single use only, cleaning of devices is strictly prohibited by IMD or Hapiguard
- If there are any questions or concerns with set up or product quality, change HapiGuard or contact customer service.
- If instructions not inside shipping package, please contact Inventive Medical Device Customer service
- Each patient may have different lengths of time to wear hapiguard. Do not exceed 5 days use for any single Guard/Patient
- Any unauthorized use of HAPIShield will void the Cap's warranty.
- Washing, cleaning, cutting or altering the mask with cleaning chemicals or instruments will make affect Hapiguard function and will no longer supported by IMD or Hapiguard.
- Please use caution in using external chemicals, Gels, Dyes or stringent hair products which will make the HapiGuard void of warranty.
- Any unauthorized use of Hapiguard with make CAP no longer supported by Warranty
- Hapiguard will not be liable for any damages for defects that may be associated with ETT, OG or NG device malfunction
- Hapiguard will not be liable if product placed into use past indicated storage life, shown on product
- Only expert clinical judgment should be used when attaching Hapiguard to a patients face and head.
- Oil-based or petroleum lubricants should never be used with oxygen delivery devices or Hapiguard.
5.Adverse Reactions : The following Adverse reactions may occur with Hapiguard
- Skin Abrasion from moving product harshly against skin
- Overtight or incorrect sizing of HapiGuard
- Patients that are not alert or wake must be checked regularly to check sizing and skin integrity.
- Clinical Staff need to move the ETT, OG/NG tube appropriately by IMD instructions


